Clinical Data Management training built for a sharper, job-ready future.
Build a CDM career with structured training in EDC workflows, query handling, data review, CDISC orientation, and interview preparation.
CDM Foundation
Learn how trial data moves from CRF design and entry to review, cleaning, reconciliation, and database lock.
Hands-on EDC Thinking
Understand how EDC environments support data entry, edit checks, discrepancy management, and audit-ready workflows.
Standards and Quality
Build context around CDISC structure, controlled terminology, and disciplined documentation used by regulated teams.

Program Architecture
A premium, single-domain learning path for Clinical Data Management.
The program is structured to reduce ambiguity. Learners start with the role of CDM in trials, then move into EDC logic, query management, standards awareness, and interview-level articulation.
What you learn
Clinical data flow, review logic, discrepancies, standards orientation, and documentation thinking.
How you practice
Through EDC-style workflow understanding, review scenarios, trackers, and process-first exercises.
Where it applies
CROs, sponsors, biotech operations, research support teams, and data-facing trial functions.

Live Online CDM Program
Hands-on EDC & CDISC training by industry experts
CDM Tools & Skills
The toolset focus is narrow by design.
This section stays aligned to Clinical Data Management operations, standards, and data review habits instead of mixing unrelated modules.
EDC Systems
Build comfort with modern electronic data capture workflows used for subject data entry, review, queries, and lock readiness.
CDISC Standards
Understand the purpose of standards such as SDTM-oriented structure, metadata discipline, and submission-friendly consistency.
SQL Basics
Develop foundational querying logic for filtering, reviewing, and validating structured clinical datasets with confidence.
Excel for Data Handling
Use formulas, review sheets, trackers, and reconciliation support techniques that remain practical in real CDM work.
MedDRA Awareness
Learn where standardized medical terminology supports coding, review, consistency, and downstream safety-related data quality.
AI-Enabled Tools
Think through edit checks, discrepancy logic, data review patterns, and exception handling with a quality-first mindset.
Tools You Will Master
Hands-on experience with industry-standard software.
Build practical proficiency with the tools that CDM teams use every day across CROs, sponsors, and research organizations.
MedDRA
Medical terminology coding
Excel
Data handling & reconciliation
PowerPoint
Reporting & presentations
ChatGPT
AI-assisted documentation
Professional communication
Program Highlights
Five comprehensive modules designed around real CDM job requirements.
Not a generic certification. End-to-end, job-oriented Clinical Data Management training designed to match real CRO and pharma hiring expectations.
Strong Theoretical Foundation in Clinical Data Management
Build a solid conceptual base through structured Clinical Data Management training delivered by industry experts with real-world experience in clinical trials and data operations.

Hands-On CDM Software & EDC Training
Move beyond theory with practical, workflow-based Clinical Data Management training using real software environments and industry-aligned processes.

CDISC Standards & Regulatory-Compliant Data Handling
Gain practical understanding of global data standards required for regulatory submissions through structured Clinical Data Management training.
AI-Enabled CDM Training & Advanced Analytics
Upgrade your skillset with India's only AI-enabled Clinical Data Management training, integrating advanced technology and smart data analytics tools.

Career Readiness, Placement Support & Interview Preparation
Convert your Clinical Data Management training into real job opportunities with structured career support.

Career + Placement
Built to help learners translate CDM training into job conversations.
The placement layer is designed around credibility: workflow fluency, clearer resume positioning, and more confident interview explanations.
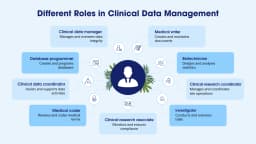
Clinical Data Coordinator
CDM Associate
Database Support Executive
Data Review Trainee
Query Management Analyst
EDC Operations Support

Placement Visual

Why This Program Stands Out
AI-enabled CDM training — end-to-end, job-oriented, pharma-aligned.
Not a generic certification. Every module is built to match real CRO and pharma hiring expectations with hands-on software exposure and strong regulatory grounding.
Only Clinical Data Management
The program removes CR, PV, RA, and medical coding distractions so learners build sharper CDM depth.
Workflow-first learning
Every module is anchored in how teams actually handle review, discrepancies, quality checks, and delivery pressure.
Standards-aware curriculum
Learners get exposure to EDC logic, CDISC orientation, and terminology discipline expected in regulated environments.
AI-enabled review mindset
The program shows where AI tools assist CDM work, where human judgment is irreplaceable, and how to use both effectively.
Interview and placement support
Preparation is tied to job conversations, role expectations, and practical explanations recruiters expect to hear.
Built for allied health graduates
Friendly for learners moving into CDM from pharmacy, nursing, biotech, physiotherapy, and life sciences.
Who Is This Program For
Designed for pharmacy and allied health graduates entering CDM.
Tailored for graduates looking to build a focused, job-oriented CDM career from a strong scientific base.

B.Pharm
Strong fit for graduates who already understand medical language, therapeutic context, and regulated documentation.

M.Pharm
Advanced pharmaceutical knowledge for specialized data roles in CROs and sponsor companies.

Nursing
Clinical exposure and patient-care understanding translates well into trial data review and medical coding functions.

Biotech
Scientific background enables deeper understanding of study design, endpoints, and biological data quality.

Physiotherapy
Allied health graduates bring practical clinical knowledge applicable to musculoskeletal and rehabilitation trial data.

Life Sciences
Broad scientific foundation prepares graduates for cross-functional CDM roles at CROs and sponsors.

B.Pharm
Strong fit for graduates who already understand medical language, therapeutic context, and regulated documentation.

M.Pharm
Advanced pharmaceutical knowledge for specialized data roles in CROs and sponsor companies.

Nursing
Clinical exposure and patient-care understanding translates well into trial data review and medical coding functions.

Biotech
Scientific background enables deeper understanding of study design, endpoints, and biological data quality.

Physiotherapy
Allied health graduates bring practical clinical knowledge applicable to musculoskeletal and rehabilitation trial data.

Life Sciences
Broad scientific foundation prepares graduates for cross-functional CDM roles at CROs and sponsors.
Free Resource
Download the full CDM syllabus PDF
Module-by-module breakdown of every topic, tool, and outcome — including CDISC, EDC, and placement-prep timeline.
FAQ
Clear answers to questions learners ask before they commit.
Honest, CDM-specific answers to help you make an informed decision about your next career step.
Apply Now
Start your CDM application with a faster, cleaner enquiry flow.
Fill in your details and submit. Our team will review your enquiry and reach out within one business day.
Contact Us
Talk to Medsafe Solutions about the CDM program.
Reach out for eligibility, batch timing, career pathway questions, or a direct discussion about how this program fits your background.
Registered Office
19-27-43/1, Sampath Niwas Apartment, Near Gandhi Nagar Park, Pedagantyada, Visakhapatnam, Andhra Pradesh 530044, India
Mon–Sat · 9:30 AM – 7:00 PM
Founder: Chintapenta Santosh
Classes are conducted online.
Prefer to talk to us now?
Call or message us directly — Mon–Sat · 9:30 AM – 7:00 PM.